What is eFSAP?
Introduction to eFSAP
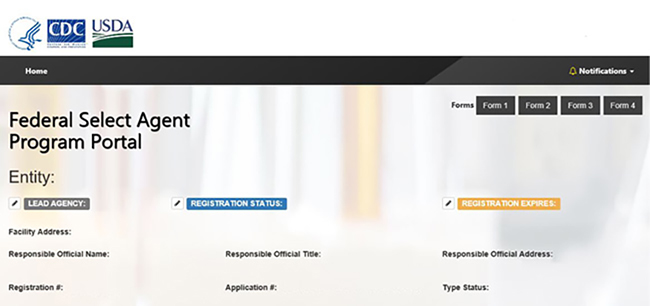
FSAP’s new secure information system, eFSAP, is used to submit select agent program information. For the first time, this two-way portal will be accessible by both FSAP and the regulated community. The new system will increase efficiency by greatly enhancing information exchange with FSAP and collaboration.
For users at registered entities, benefits of the system include reduced paperwork, increased ease of validating and submitting information (via use of the electronic portal), and reduced processing time for requests (as real-time information exchange will allow for increased responsiveness).
Access
To gain access to the system, all users are required to establish a SAMS (Secure Access Management Services) account, which protects the security of the information by requiring users to enter a user ID and password, or other secure credentials, before providing access to the system. Users with SAMS accounts will only have access to their own entity’s information. If you need assistance with this, please contact us.
Functionality
More information on these forms can be found here.